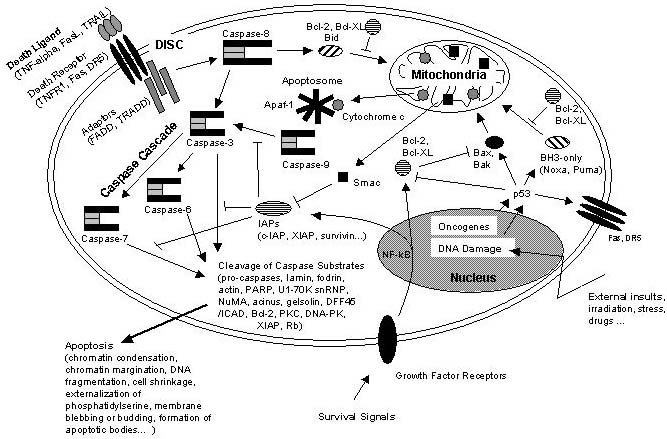
Figure 5
Schematic representation of some major apoptotic signalling pathways.
Apoptosis can be induced in response to various signals from inside and outside the cell, e.g. by ligation of so called death receptors or by cellular stress triggered by oncogenes, irradiation or drugs. Signals emanating from death receptors initially activate the Death Inducing Signalling Complex (DISC) which mediates activation of the initiator caspase-8. Activated caspase-8 initiates a caspase cascade by processing the effector caspases-3, -6, and – 7 which in turn cleave a number of protein substrates. Cleavage of caspase substrates eventually leads to the characteristic morphological and biochemical features of apoptosis. In some cell systems, this direct caspase cascade is sufficient to elicit apoptosis on its own (type 1 signalling), whereas in other cases the signal coming from the DISC must be amplified by the proteolytic activation of the BH3-only protein Bid by caspase-8 with subsequent induction of apoptotic events at the mitochondria (type 2 signalling). Mitochondrial apoptotic signalling includes the release of cytochrome c from the mitochondrial intermembrane space to the cytosol where it contributes to the formation of the apoptosome which consists of cytochrome c, Apaf-1 and dATP. The apoptosome activates caspase-9 which is another initiator caspase and thus is able to mediate the caspase cascade by activating caspase-3. Another mitochondrial proapoptotic factor is Smac which acts by inhibiting the IAPs from blocking caspase activity. IAPs are a family of proteins with antiapoptotic activity by directly inhibiting caspases. IAP expression can be upregulated in response to survival signals such as those coming from growth factor receptors, e.g. by activation of the transcription factor NF-kB, therefore providing a means to suppress apoptosis signalling. Of central importance are the antiapoptotic Bcl-2 family members such as Bcl-2 and Bcl-XL which counteract the action of BH3-only proteins such as Bid but also of proapoptotic Bax and Bak and thus can inhibit mitochondrial proapoptotic events. Apoptotic signals coming from the inside of the cell frequently have their origin within the nucleus, being a consequence of DNA damage induced by irradiation, drugs or other sort of stress. DNA damage in most cases eventually results in the activation of the p53 transcription factor which promotes expression of proapoptotic Bcl-2 members and suppresses antiapoptotic Bcl-2 and Bcl-XL. Other organelles besides mitochondria and the nucleus, such as the ER and lysosomes also have been implicated in apoptotic signalling pathways, and it should be kept in mind that presumably hundreds of proteins are part of an extremely fine-tuned regulatory network consisting of pro- and antiapoptotic factors.
Schematic representation of some major apoptotic signalling pathways.
Apoptosis can be induced in response to various signals from inside and outside the cell, e.g. by ligation of so called death receptors or by cellular stress triggered by oncogenes, irradiation or drugs. Signals emanating from death receptors initially activate the Death Inducing Signalling Complex (DISC) which mediates activation of the initiator caspase-8. Activated caspase-8 initiates a caspase cascade by processing the effector caspases-3, -6, and – 7 which in turn cleave a number of protein substrates. Cleavage of caspase substrates eventually leads to the characteristic morphological and biochemical features of apoptosis. In some cell systems, this direct caspase cascade is sufficient to elicit apoptosis on its own (type 1 signalling), whereas in other cases the signal coming from the DISC must be amplified by the proteolytic activation of the BH3-only protein Bid by caspase-8 with subsequent induction of apoptotic events at the mitochondria (type 2 signalling). Mitochondrial apoptotic signalling includes the release of cytochrome c from the mitochondrial intermembrane space to the cytosol where it contributes to the formation of the apoptosome which consists of cytochrome c, Apaf-1 and dATP. The apoptosome activates caspase-9 which is another initiator caspase and thus is able to mediate the caspase cascade by activating caspase-3. Another mitochondrial proapoptotic factor is Smac which acts by inhibiting the IAPs from blocking caspase activity. IAPs are a family of proteins with antiapoptotic activity by directly inhibiting caspases. IAP expression can be upregulated in response to survival signals such as those coming from growth factor receptors, e.g. by activation of the transcription factor NF-kB, therefore providing a means to suppress apoptosis signalling. Of central importance are the antiapoptotic Bcl-2 family members such as Bcl-2 and Bcl-XL which counteract the action of BH3-only proteins such as Bid but also of proapoptotic Bax and Bak and thus can inhibit mitochondrial proapoptotic events. Apoptotic signals coming from the inside of the cell frequently have their origin within the nucleus, being a consequence of DNA damage induced by irradiation, drugs or other sort of stress. DNA damage in most cases eventually results in the activation of the p53 transcription factor which promotes expression of proapoptotic Bcl-2 members and suppresses antiapoptotic Bcl-2 and Bcl-XL. Other organelles besides mitochondria and the nucleus, such as the ER and lysosomes also have been implicated in apoptotic signalling pathways, and it should be kept in mind that presumably hundreds of proteins are part of an extremely fine-tuned regulatory network consisting of pro- and antiapoptotic factors.